

Paradigm Shift Intervention Monitoring
 twitter
twitter

Commentary
West Virginia
Reports H3N2v and Seasonal H3 in Week 32
Recombinomics
Commentary 13:00
August 30, 2012
Initial
testing of the specimen indicated a seasonal influenza A (H3N2) virus
and the specimen was submitted to CDC as a routine surveillance sample.
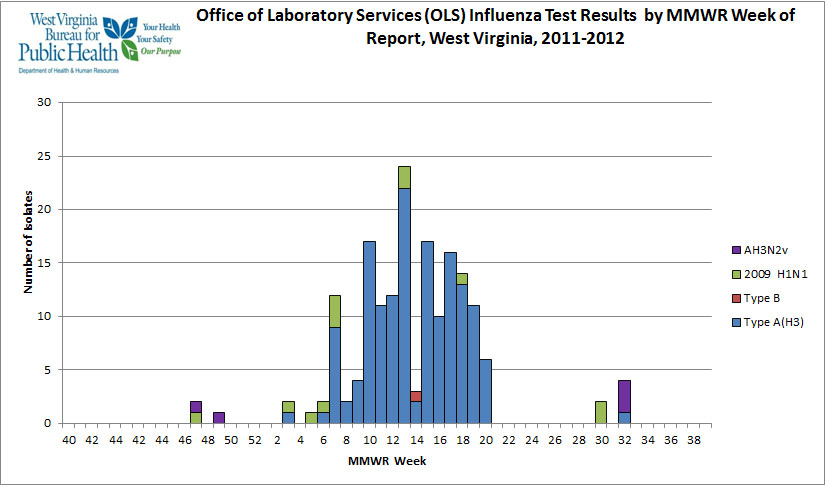
The above graph
displays West Virginia serotypes in the 2011/2012 season. The
first two examples of the current H3N2v sub-clade sequences (A/West
Virginai/06/2011 and A/West Virginia/07/2011) are displayed in the
above graph as are the three 2012 H3N2v cases (week 32) who were
reported to have attended the Gallia Junior County Fair in Ohio.
Gallia County ran rapid tests on 200
symptomatic attendees and 69 were influenza A positive. 11 of
the cases were listed in the Ohio Department of Health report of August
13. In the August 24 report, the number of confirmed cases
increased to 12, raising concerns of new cases.
The hundreds of H3N2v cases in
Gallia County, Ohio led to local
testing of children in Ashland County, Kentucky, which is adjacent
to Galiia. Influenza A positive samples from six cases were sent
to the Kentucky State lab where they were diagnosed as “seasonal” H3n2
as noted in an Ashland-Boyd
press release and a Kentucky health alert.
West Virginia also reported a
seasonal H3 cases in week 32, as seen in the above graph, which also
has 3 H3N2v cases reported in the same week. The West Virginia
seasonal H3 samples was not sent to the CDC for confirmation, and
Kentucky and the CDC failed to respond to repeated inquiries on CDC
confirmation of the six cases in Kentucky.
The above comments
describe the testing of A/Pennsylvania/40/2010, which was initially
reported as seasonal H3. This mis-diagnosis was also reported for
A/Maine/07/2011 as well as A/West Virginia/07/2011
because the RNA level in the 2011 cases was low, so the CDC RT-PCR test
yielded a weak
H3 positive and a negative on the H1N1pdm09 NP target which is used
to identify H3N2v cases which are H3 positive. The West Virginia
2011 contact was intially reported
as influenza A. In the 2011 cases H3N2v dianosis was based on
partial sequencing. Full sequences for these recent cases have
never been reported, because the low level of RNA precluded
culturing. The 2010 case was eventually cultured, but the
sequence was reported 5 months after collection.
Thus, samples which are
classified by seasonal H3 by RT-PCR require sequence confirmation,
especially if the RNA levels are low, yet there have been no statements
from the CDC, Kentucky, or West Virginia that the August “seasonal” H3
cases in counties adjacent to the Gallia H3N2v outbreak have been
sequence confirmed, even though the CDC has asked physicians to
“consider” testing off season ILI cases with no swine contact.
The West Virginia week 32
seasonal H3 was not sent to the CDC because the CDC did not ask for the
sample.
The
heavily biased CDC testing of ILI with swine contact continues to be
hazardous to the world’s health.
Media
Link
Recombinomics
Presentations
Recombinomics
Publications
Recombinomics
Paper
at Nature Precedings